© A.W.Marczewski 2002
A Practical Guide to Isotherms of ADSORPTION on Heterogeneous Surfaces
Global Heterogeneity, H:
calculation plot: GL
(see local heterogeneity h(θ) function)
Global heterogeneity plot θ(1-θ) vs. ln(p) for Generalized Langmuir (GL) isotherm and its special cases (Langmuir-Freundich, LF, Generalized Freundlich /Sips, GF, and Langmuir-Freundlich, LF) compared with Langmuir equation.
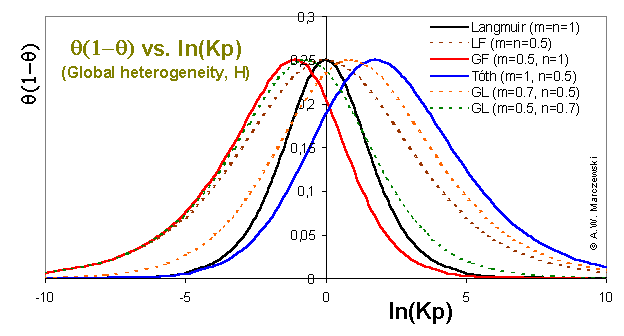
The position of the "peak" maximum corresponds to the θ = 0.5, i.e. half-filled monolayer. All plots should be compared to Langmuir isotherm line for the same average energy:
Kavg = Koexp(Eavg) where Eavg = εavg/RT
or for asymmetric energy distributions like GF and Toth, for the characteristic energy Ee (e.g. for GF, Ee = Emin)
Ke = Koexp(Ee) where Ee = εe/RT
which is the reference for monolayer localized physical adsorption - if no lateral effects are present the isotherm "peak" in this plot should always be wider than the Langmuir. Another possiblity of comparison is shifting isotherms in such a way that their θ = 0.5 points ("peak" max.) coincide.
Higher extension to the right (higher pressures are required for adsorption) of the maximum corresponds to the heterogeneity at low energies (for energies below average energy), whereas the extension to the left (adsorbate adsorbs at lower pressures than for Langmuir iso.) corresponds to the heterogeneity at high energies (higher than the average energy).
Systems characterized by similar heterogeneity parameters m corresponding to energy distributions extended towards higher adsorption are all very similar for low coverages. Likewise, the behavior of isotherms for high coverages is similar (though the lines are shifted on the ln(p) axis) if their heterogeneity parameters n (corresponds to low energy range) are similar. For lower values of parameters m, n the plot "peak" beacomes wider evidencing higher heterogeneity. The most narrow "peak" is for homogeneous Langmuir isotherm.
NOTE. Replace p with c for dilute solute adsorption.
Send a message to Adam.Marczewski AT@AT umcs.lublin.pl